Let’s explore endothermic reactions! Most people have tried doing a baking soda and vinegar chemical reaction, but did you know that this reaction is actually COLD?
I had no idea until we studied endothermic and exothermic reactions in my son’s 8th grade science book and learned that baking soda and vinegar is an endothermic reaction that absorbs heat from its surroundings. It’s so cool to measure how cold the products of this reaction get. Literally!

What is an endothermic chemical reaction?
During a chemical reaction, more energy is required to break bonds between atoms than to form bonds. When bonds are broken, energy is absorbed (because energy from the surroundings were needed to have enough to break the bonds), and when bonds are formed, energy is released or given off. In a given chemical reaction, bonds may be broken and new bonds formed. One would need to look at the energy absorbed and the energy released to see which is greater.
If more energy is released than the energy that is absorbed, it is an exothermic reaction and feels warm. If more energy is absorbed than released, it is an endothermic reaction and feels cold.
Winter is a great time to explore endothermic reactions, although this is fun to do anytime.
This post contains Amazon affiliate links, which means that I earn from qualifying purchases at no additional cost to you.
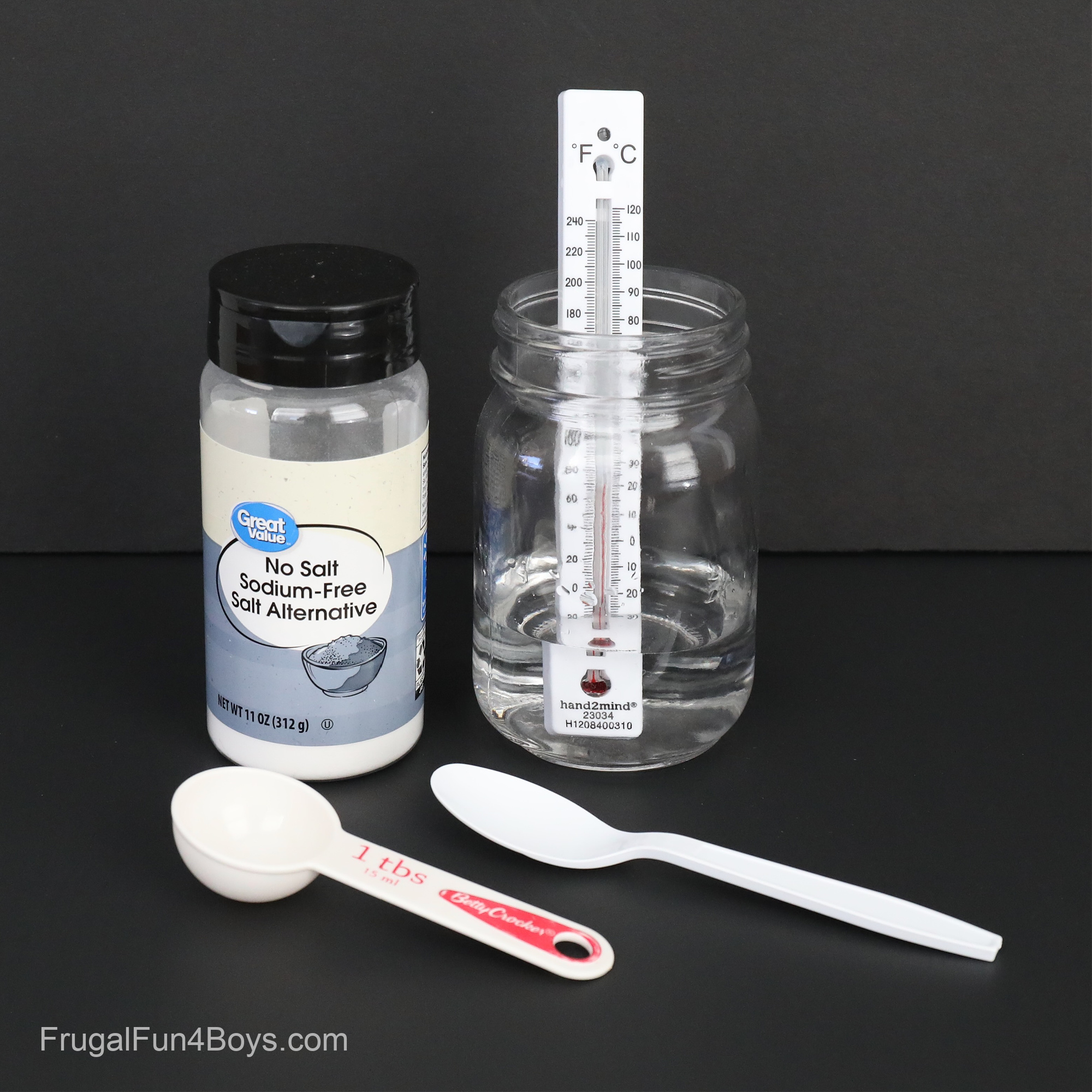
Supplies Needed for a Baking Soda and Vinegar Endothermic Reaction:
- A jar
- A pie plate or something similar
- A thermometer – we used these thermometers from Amazon
- Baking soda
- Vinegar
- Liquid dish soap
- Food coloring (optional)
- Glitter (optional)

Start by pouring some vinegar into your jar. The amount needed will depend on the size of the jar, but filling it about 1/4 full of vinegar is plenty.
We added food coloring, glitter, and dish soap just to make the reaction bigger and more pretty. When the baking soda and vinegar react, the dish soap will create lots of foam that will last longer than the bubbles from the reaction.
The glitter just gives it a sparkly, wintery look!

Stir up the vinegar, food coloring, and glitter. Then check the temperature of the liquid.
Ours was 66 degrees.

Dump in 1 Tablespoon of baking soda, and watch what happens!
My 8 year old was so impressed with all the bubbles and foam.

It just gets bigger…

And bigger!

Then check the temperature. Make sure that the bottom of the thermometer is still sitting inside the liquid in the bottom of the jar.
Ours was 59 degrees! The reaction absorbed heat and caused the temperature to drop 7 degrees. The outside of the jar felt cold.
Out of curiosity, I tried reacting ONLY baking soda and vinegar to see if the dish soap was reducing the temperature drop. With baking soda and vinegar alone, the temperature dropped 9 degrees. Super cool!!

Other Endothermic Chemical Processes:
Try dissolving salt substitute (potassium chloride) in water!
This reaction is not very exciting to watch. However, the temperature will drop 10 – 12 degrees and the jar feels quite cold. So do the baking soda and vinegar one first, and then try this reaction for a larger temperature drop!
Need more winter science ideas?
Here are more than a dozen fun Winter STEM Activities. Experiment with static electricity, design a container that keeps ice from melting, learn why arctic animals have large paws, and lots more.


0 Comments
Post a Comment